Why Water is Weird
Why Water is Weird
Most people understand certain physical concepts well enough to be permanently confused by them. This is especially true when the subject involves the inter-molecular dynamics of the H2O molecules to other H2O molecules. In two previous posts on this website I discussed the molecular basis for non-Newtonian fluids. If you had read these posts and spent some time studying them you would understand the quirkiness of the H2O molecule that underlies non-Newtonian fluids and other quirky characteristics of water. Did anybody do this? Of course not. That is because, like I said, most people understand certain physical concepts well enough to be permanently confused by them. And when people become confused their minds fog over and they lose interest. Here is your second chance.
As discussed therein, the reason the hammer blow on the surface of the corn starch and water mixture caused the first of the two hydrogen bonds to be broken was because the high granularity of corn starch was able to apply force between water molecules that separated the very weak hydrogen bond that existed between the water molecules. And, the reason the corn starch mixture instantly became strong (hard) is because of the strength of the hydrogen bonds that existed between the water molecules. Did you notice anything contradictory about these last two sentences? If not, read them again. In the first sentence I said the hydrogen bond is weak in the second I said the hydrogen bond is strong. How is that possible, you might be wondering. How can a hydrogen bond be weak in one instance and strong in another? Did I, maybe, make a mistake?
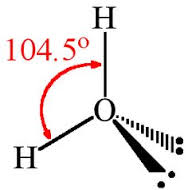
I did not make a mistake. Water’s hydrogen bond is both weak and strong. How is that possible? There are two peculiarities about the water molecules’s hydrogen bond that you need to understand. Firstly, the oxygen side of each water molecule comprises the negatively charged end of its polarity. On this negatively charged, oxygen location there are two attachment points at which a positive, hydrogen atom of two other water molecules can form a hydrogen bond. It’s important to be cognizant of the fact that, in sharp contrast to the positive, hydrogen side of the water molecule which are far apart from each other, on this negative side the two attachment points are very close to each other. As I will explain shortly, understanding the implications of the fact that these two negative locations are close to each other is THE MOST IMPORTANT CONCEPT to understand if you want to understand the collective quirkiness of H2O molecules.
But before we get to that there is another concept that you have to understand. Unlike either a covalent bond or an ionic bond the forces that underlie a hydrogen bonds are neutralized with the completion of a hydrogen bond. This is because the force that underlies a hydrogen bond is an implication of the polarity of the H2O molecule itself–the fact that it is geometrically out of balance. With the completion of both hydrogen bonds that balance is restored and the force is neutralized.
You should now have a firm grasp of these two concepts: 1) the fact that the the two negatively charged hydrogen bonding attachment points on each water molecule are close together on its oxygen molecule and, 2) the fact that the completion of the hydrogen bond neutralizes (consumes) the force that established the hydrogen bonds.
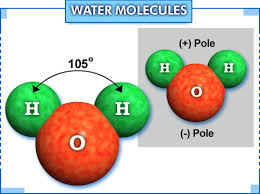
(At this point in time it might be a good idea to get out a piece of paper and draw a rough sketch of a water molecule and put a dot at each of the four attachment points. Use the picture of the H2O molecule at the top of the page as your model. Be sure to maintain its orientation. Put one dot on each of the two hydrogen molecules and put two dots, close to each other, at the far end of the oxygen molecule. Enumerate these dots, starting at the top left, going left to right, ending with the last dot on the bottom. Take note of the fact that the distance between #1 and #2 is about 4 times that between #3 and #4. In accordance with the nomenclature indicated in the video indicated below write +1∂ on each of the two hydrogen atoms and -2∂ near the bottom of the Oxygen atom.)
Now you have all the elements you need to complete the final step, which involves asking yourself some rhetorical questions about the implications of 1 or 2 hydrogen bonds having been completed. What it comes down to is this: when two of the hydrogen bonds are completed (on the negative end of a water molecule) the bonds are extremely weak. It’s polarity is neutralized and the resulting force of the bond disappears (2∂ – 2∂ = 0∂). This is a result of having its balance restored by the presence of four bonds around its perimeter (two covalent bonds and two hydrogen bonds). Consequently the two hydrogen bonded atoms (the ones associated with the adjacent water molecules) just kind of float. They maintain a very weak, ephemeral presence. The only thing holding them is the partial return of the polarity whenever they move away.
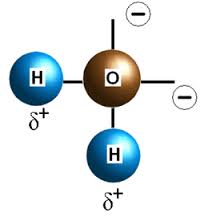
Now ask yourself what happens when you pull one of them away completely. When only one hydrogen bond is complete the bond strength is relatively high. This is because, unlike when there are two, when there is only one hydrogen bond on the negative, oxygen location the polarity has not been fully consumed. Its force has not been fully neutralized (2∂ – 1∂ = 1∂).
This results in a bond that is structurally very strong. This is the molecular basis for the collective quirkiness of the H2O molecule. It is structurally stronger (higher viscosity) when it is less interconnected (less self attached) and less dense (as in ice) and structurally weaker (lower viscosity) when it is more interconnected (more self attached) and more dense (as in liquid water).
At this juncture it might be a good idea to re-examine two posts that I pointed you at previously:
What is the molecular mechanism underlying non-Newtonian fluids?
http://wp.me/p4JijN-46Z
Solution to the molecular mechanism underlying non-Newtonian fluids?
http://wp.me/p4JijN-48z
Take your time with these. It’s very important that you fully comprehend what is going on here.
More concisely, this mechanism dictates that the first of the two bonds (associated with the negatively charged, oxygen side of a water molecule) can be separated very easily, the second cannot. Consequently, there is a structural strength hidden from us in water molecules that only becomes evident the more they are detached from each other. One of several counter intuitive implications of this mechanism is that clusters/droplets of H2O can be separated from each other using relatively low energy, as in evaporation, but relatively high energy levels are required to achieve total separation, as in gaseous H2O (steam). (Also, the widespread, mistaken perception that evaporation produces gaseous H2O [steam] is further bolstered by the fact that the small clusters of H2O evaporate [vapor] is, often, equally as invisible as is steam.)
There is another gigantic implications associated with the hidden strength of water’s hydrogen bond. It involves water droplets/clusters at wind-shear boundaries being bombarded with side-glancing impacts from N2 and O2 and beginning to spin and elongate into chains (polymers of H2O joined by strong hydrogen bonds [2∂ – 1∂ = 1∂]) as a result of the centrifugal force produced by the spinning. This is the basis of the plasma that comprises the structure of vortices. (Its a plasma because the forces pulling the H2O molecules apart from each other [the centrifugal force that is a result of the spinning caused by wind shear] are opposed by the forces pulling it back together again (electro-magnetic forces of polar molecules [2∂ – 1∂ = 1∂]). These vortices are the conduits of low pressure energy in our atmosphere (as in jet streams, storms, tornadoes, and hurricanes). When you understand this you will understand something extremely profound about the nature of atmospheric flow.
Vortex Phase: The Discovery of the Spin that Underlies the Twist
Vortex Phase: The Discovery of the Spin that Underlies the Twist

too glossed over. add detail so it is believable
it is a pile of steaming poop
11/22/14
James McGinn, Mr Solving Tornadoes,
RETRACTS HIS BOOK “Solving Tornadoes”
……………………………..
from his previous post;
“Well, let’s just say that it ain’t no textbook.
The confusion you are feeling is by design. And it’s not so
simple as me having an objection to pandering to my audience.
It’s more like I recognize that most consumers of science are
looking for an excuse not to think. I wrote it with with the
intention of giving you no avenue of escape.
You can try to dispute it if you want. But, let’s face it,
you’re already entangled. If you struggle you’ll just become
more entangled. And there’s no way out — without thinking.
Sorry.
My only advice is that if you haven’t gotten to the third
chapter yet stop now. But it sounds like it’s too late for
that. Oh well. You had a good run. And it’s not like me
making you think is some kind of violation of your civil
rights or something. ”
…………………………….
don’t bother to reply to this guy, **ALL** marbles are loose, the cheese
slid off his cracker, and he is only here to argue with anyone.
Jim McGinn in his Book “Solving Tornadoes: Mastering the Mystery of the
Vortex” represents ‘the beginning of the end’ of what little
rationality mankind was capable of applying to any given subject. McGinn
represents an unusually high order of the Dunning-Kruger effect seldom
seen in ‘modern’ internet work; What other authors are to Chemtrails and
HAARP mind control Jim McGinn is to understanding (or mis-understanding)
tornadoes …
To cite Jim McGinn as an idiot or a moron is to slight idiots and morons.